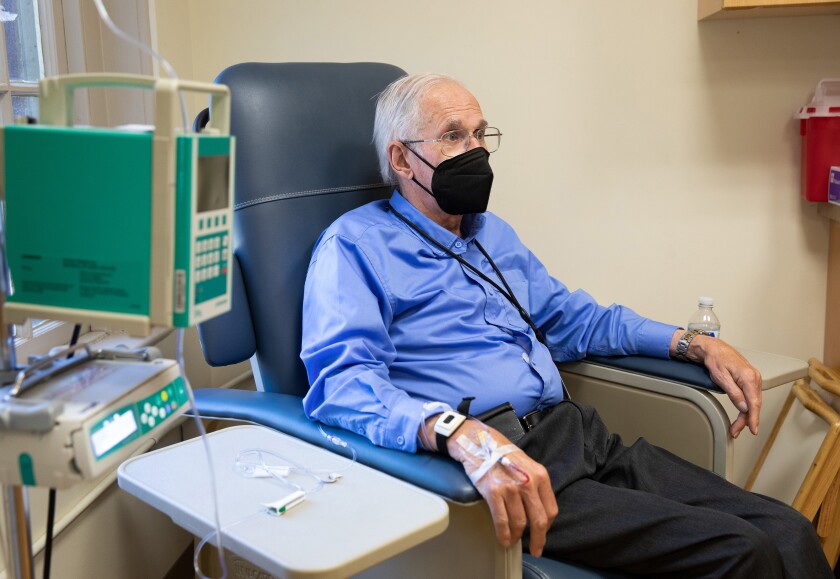
The Meals and Drug Administration’s shock approval of Aduhelm for the therapy of Alzheimer’s illness final yr was a multitude on virtually each stage. Three company advisors resigned, and skeptical medical doctors comparable to myself have been left to advise sufferers — all determined for hope — that, sure, it's a therapy choice however, no, we don't know whether or not it's going to work.
And by the best way, it's terribly costly.
This isn't the place you wish to be when coping with this mysterious and relentless illness that takes an individual’s life solely after stripping away reminiscences and each different vestige of who we're as people.
However this seemingly rash act by the FDA, adopted by a sudden retreat by the Facilities for Medicare & Medicaid Providers final month to cowl Aduhelm’s use solely in medical trials, could find yourself having an unexpected and long-lasting profit. As a result of this was the primary drug ever prescribed to combat the development of Alzheimer’s, it revealed simply how a lot work the medical group nonetheless must do to organize itself to deal with Alzheimer’s sufferers, not simply research them.
We have to create a sprawling infrastructure — a complete medical tradition, frankly — to accommodate and assist the roughly 6 million Individuals with Alzheimer’s. And we had higher do it quick, as a result of a number of promising medicine are wending their method by means of the approval course of.
Simply as COVID-19 confirmed us how society was not prepared for a pandemic, however ready us for the following one, so too has Aduhelm proven us what must be executed to organize for the following Alzheimer’s drug. It was, in a way, a trial run.
What did we be taught? We don’t have sufficient clinicians who specialise in Alzheimer’s or medical doctors with neurological experience. We don’t have panels to find out therapy choices, comparable to hospital multispecialty boards do for most cancers sufferers. We don’t have sufficient infusion facilities to offer sufferers these medicine, which like Aduhelm will more than likely need to be delivered intravenously. We don’t have transportation methods to get sufferers with out means to those facilities commonly.
We aren't prepared. However we’ve bought to be.
Aduhelm was by no means promised to be a treatment. Relatively, the drug is designed to halt and reverse the expansion of sticky amyloid plaques within the mind which can be thought by many to trigger Alzheimer’s debilitating results. It seeks to flatten the curve of signs, to make use of a pandemic-era phrase.
How do you measure its success? When is the price — simply lowered to $28,000 a yr, from $56,000 — value it? Who decides?
Aduhelm’s approval despatched us all scrambling. These of us who’d been following its tortured journey — in 2019, for instance, trials have been stopped as a result of it confirmed no indicators of working — by no means imagined that we’d need to activate a dime and get all the required elements in place to get it prescribed to everybody who was eligible for it; at UCLA Well being, this meant fairly actually tens of 1000's of individuals.
We needed to pivot, as many different facilities did, from an educational observe to a group observe virtually in a single day. This was particularly difficult as a result of many major care medical doctors aren't certified to deal with these sufferers and this infinitely complicated illness.
As a result of there has by no means been any therapy for this illness, the medical doctors within the subject at this time got here to do analysis. With the approval of Aduhelm, quite a lot of lab scientists discovered themselves making ready to do much more medical work than they signed up for. We have to begin a pipeline of certified Alzheimer’s clinicians now, as a result of extra therapies are coming.
The brand new medicine on the horizon are additionally designed to sluggish the unfold of amyloid plaques. They're donanemab from Eli Lilly, lecanemab from Eisai/Biogen and gantenerumab from Roche/Genentech.
Eisai/Biogen has already utilized for a advertising and marketing license, citing information from its Part II trial that present robust plaque discount. The opposite two firms are anticipated to observe swimsuit quickly. Though within the early levels of trials, a number of of those medicine could nicely show more practical than Aduhelm, often known as aducanumab.
After all, analysis should proceed. These medicine don’t fake to be cures with the power to undo injury. We'll nonetheless want all of the lab scientists and devoted medical researchers we are able to get to search out methods, lastly, to stop and reverse the illness.
Within the early days of COVID-19, hospitals have been caught with out sufficient protecting gear, sufficient ICU beds and sufficient ventilators. Everybody was operating on intuition. By the point Omicron got here round, hospitals have been much better ready and knew what to do. That is one motive that there have been far fewer deaths with this newest surge of infections.
Similar goes for Aduhelm and the teachings we realized. We aren’t prepared but, however because of this trial run, we now know what we've got to do to prepare.
Keith Vossel is the director of the Mary S. Easton Heart for Alzheimer’s Illness Analysis at UCLA.
Post a Comment